By Nico Bacharidis, Maria Samoili, and Ruven Remo Eul
WHAT is Value-based tendering
Value-based tendering (VBT) in the life sciences industry marks a transformative approach to procurement, particularly in Life Sciences such as pharmaceuticals, biotechnology, and medical devices. At its essence, VBT reorients tender processes from a narrow focus on acquisition costs to a broader evaluation of value delivered to stakeholders, including patients, healthcare providers, payers, and society at large. Unlike traditional price-based tendering, where typically the lowest bidder often wins, VBT incorporates multifaceted criteria to achieve the “best value for money.” The European Public Procurement Directive EU PPD 2014/24/EU refers to this as MEAT tenders = Most Economically Advantageous Tenders. This is especially pertinent in life sciences, where products like drugs and therapies have long-term impacts on health outcomes, resource utilization, and environmental sustainability.

Tender management has surged in importance within the pharmaceutical sector over the past decade. According to industry analyses, tenders now account for about 30–40% of the global pharmaceutical market by value, up from lower figures in the early 2010s. Depending on the portfolio, especially in the areas of generics, biosimilars, vaccines or blood-plasma products, the tendering share can be significantly higher. In Medtech the importance of tendering is expected to be even higher and closer to 70%–80%. This growth is driven by escalating healthcare costs, government austerity measures, and the need for efficient resource allocation in public health systems. Projections suggest this share could continuously rise, fueled by the increasing usage of procuring specialized drugs, including biologics, oncology treatments, and rare disease therapies through tenders.
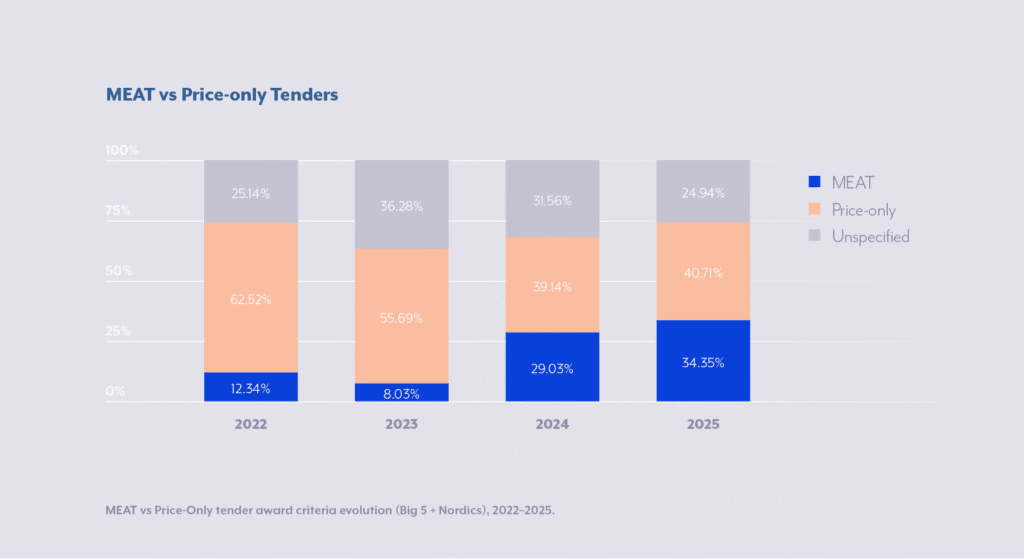
Historically, tenders were predominantly used for generic medicines to drive down prices through competition. However, as patents expire on more complex molecules and many biosimilars have entered the market, specialized manufacturers, such as those producing monoclonal antibodies or gene therapies, are compelled to engage in tender processes. This expansion poses challenges for innovators, who must balance competitive pricing with recouping R&D investments. Our recent study confirmed that purchase price typically constitutes around 70% of bid award criteria across sectors like pharmaceuticals, medtech, and diagnostics. This figure varies significantly based on the specific country, tender authority, and type of product/ product portfolio involved. Additional studies, such as those from the European Federation of Pharmaceutical Industries and Associations (EFPIA), echo this, noting that while price remains dominant, there’s a gradual shift toward value-oriented metrics in about 20–30% of European tenders.
In Europe, the revised EU Public Procurement Directive 2014/24/EU, effective since 2016, has been a catalyst for this change. The directive explicitly endorses the Most Economically Advantageous Tender (MEAT) framework, which mandates that awards be based on the best price-quality ratio rather than lowest cost alone. At its core, MEAT integrates cost considerations with qualitative elements, allowing for a more nuanced evaluation. Key components include:
- Qualitative Criteria: Technical merit, efficacy, and innovation, such as a drug’s ability to improve quality-adjusted life years (QALYs) or reduce treatment side effects.
- Social Criteria: Aspects like labor standards, diversity in supply chains, and contributions to public health equity.
- Innovative Criteria: Rewards for novel delivery mechanisms, digital health integrations, or R&D commitments that advance therapeutic areas.
- Sustainable and Environmental Criteria: Evaluation of the product’s lifecycle, including carbon footprint, waste reduction, and eco-friendly manufacturing practices.
- Other Criteria: Criteria which are often seen in pharmaceutical Tenders are supply-reliability and supply-chain security, additional services, packaging features or terms & conditions.
This holistic approach enables tender authorities to consider the total cost of ownership (TCO), factoring in long-term savings from better outcomes. For example, a higher-priced therapy might be selected if it demonstrates superior adherence rates, leading to fewer hospitalizations and overall cost reductions for the healthcare system.
While we see more and more MEAT-tenders across countries, there is still significant room for improvement. This is being reflected by latest policy developments, as in October 2025, the European Commission published a major evaluation of the Public Procurement Directives as the first step toward a full revision. Key findings relevant to MEAT and ESG:
- Green, social, and innovative procurement is progressing but uneven across the EU.
- The directives have not fully enabled strategic procurement, which includes sustainability and qualitative goals.
- Complexity and legal uncertainty reduce uptake of MEAT criteria, especially qualitative and life‑cycle requirements.
The European Commission’s Evaluation is the foundation for an upcoming reform, expected Q2 2026. And while some countries and buyers are embracing this procurement approach, many countries and tender authorities still focus solely or mostly on price. However, value-based tendering can go beyond award criteria and look at broader ways to improve the attractiveness and sustainability of the market. This can mean moving from winner-takes it all tenders to multiple winners, which diversifies the risk of supply issues and out-of-stocks across multiple suppliers. It can also involve increasing tender submission timelines from a couple of days or weeks to 2-3 months, giving suppliers more time to customize and improve their offers. It can mean moving from a very closed buying process to one that enables some interactions and allows for innovative value propositions.
WHY is value-based tendering critical for Pharma & Med-Tech companies
Globally, tender authorities are pivoting toward more sustainable and outcome-focused procurement strategies. This is evident in regions beyond Europe, such as Australia, Canada, and parts of Asia, where health technology assessments (HTAs) increasingly influence tender designs. In the U.S., value-based purchasing (VBP) under the Centers for Medicare & Medicaid Services (CMS) mirrors VBT principles by linking payments to performance metrics likereadmission rates and patient satisfaction.
From a life sciences perspective, VBT offers several advantages:
- Enhanced Patient Outcomes: By prioritizing value, tenders encourage the selection of therapies that deliver measurable improvements in health metrics, such as survival rates or quality of life.
- Fostering Innovation: Companies are incentivized to invest in R&D for differentiated products, as MEAT criteria can allocate points for unique features like personalized medicine or combination therapies.
- Sustainability Gains: Integrating environmental factors promotes greener practices, aligning with global goals like the UN Sustainable Development Goals (SDGs) and EU Green Deal initiatives.
- Risk Mitigation: For manufacturers, building strong value dossiers, supported by real-world evidence (RWE) and health economic models, can improve win rates in tenders.
- Improve Supply Reliability: Price only tendering seems to correlate with an increase in supply issues and out-of-stock situations. Lower prices de-incentivize companies to maintain safety-stock or diversify production across multiple manufacturing sites.
However, VBT is not without controversies. Critics argue that subjective criteria can introduce biases or favoritism, potentially disadvantaging smaller players. Implementation varies; in some countries, like the Netherlands and Sweden, VBT is well-advanced with tools like dynamic purchasing systems, while others lag due to administrative burdens.
There are also key challenges within the VBT processes:
- Quantification of Value: Defining and measuring non-price criteria requires robust data. For instance, pharmaceutical companies must provide evidence from clinical trials, pharmacoeconomic studies, or post-market surveillance.
- Regional Variations: While Europe leads with MEAT, other regions like Latin America often stick to price-based models due to budget constraints. In Asia, countries like South Korea are adopting hybrid approaches via their Health Insurance Review & Assessment Service (HIRA).
- Compliance and Preparation: Firms need cross-functional teams (e.g., market access, regulatory, medical, marketing and commercial) to craft compelling bids. Best practices include early engagement with tender authorities and leveraging digital tools for scenario modeling.
In short, it becomes clear that VBT is on the merge and Life Sciences manufacturers need to familiarize themselves with the various concepts, such as the European MEAT framework or the hybrid approach in South Korea. This will allow them to build a VBT Approach & Methodology for their own company to stay ahead of the evolving procurement landscape and ultimately, achieving a competitive advantage and sustainable business results.
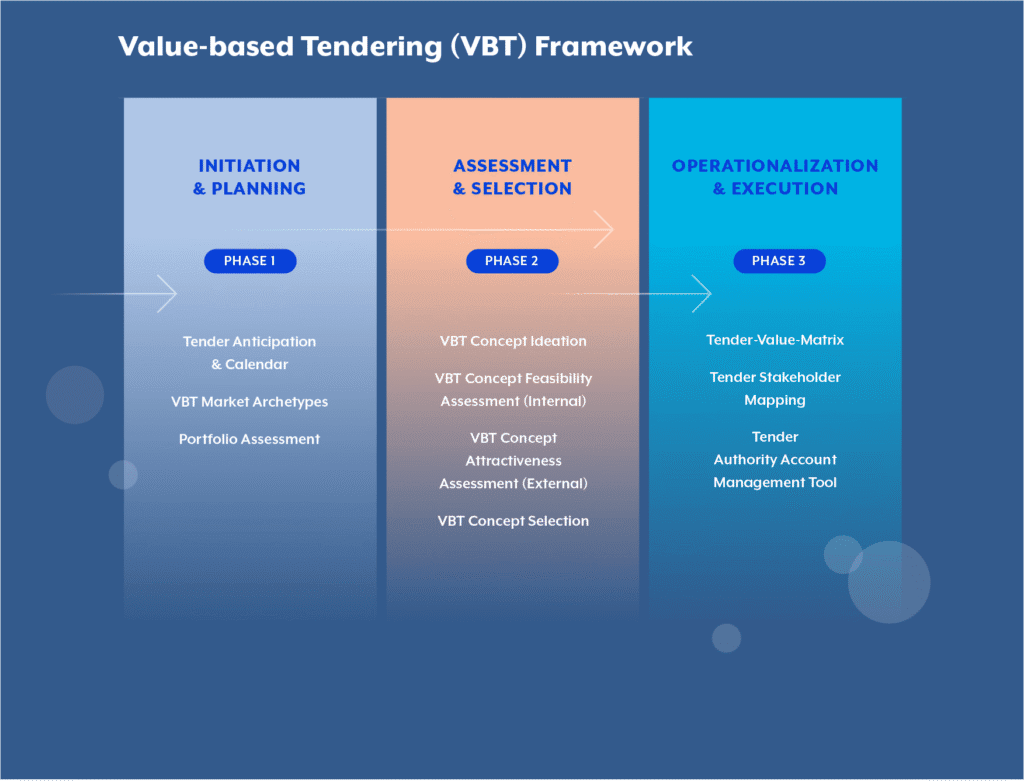
HOW is value-based tendering done
Having established the strategic importance of value-based tendering for life sciences companies, the question becomes: how can organizations systematically implement VBT to maximize their competitive advantage?

Phase 1
Initiation & Planning
Success in value-based tendering requires a methodical approach that begins with understanding where to focus efforts for maximum impact. Not every tender opportunity warrants the same level of investment in value demonstration and tender shaping activities. Companies must strategically assess their portfolio, market position, and competitive landscape to identify the opportunities where value-based approaches will deliver the greatest return on investment and ensure a sustainable and attractive market in the long term.
The portfolio assessment provides an inward-looking view, identifying which products or therapeutic areas have the differentiation, evidence, and strategic relevance to justify a value-based approach. In parallel, developing data-driven market archetypes offers an outward-looking lens, mapping how tender markets behave and evolve across geographies and procurement authorities.
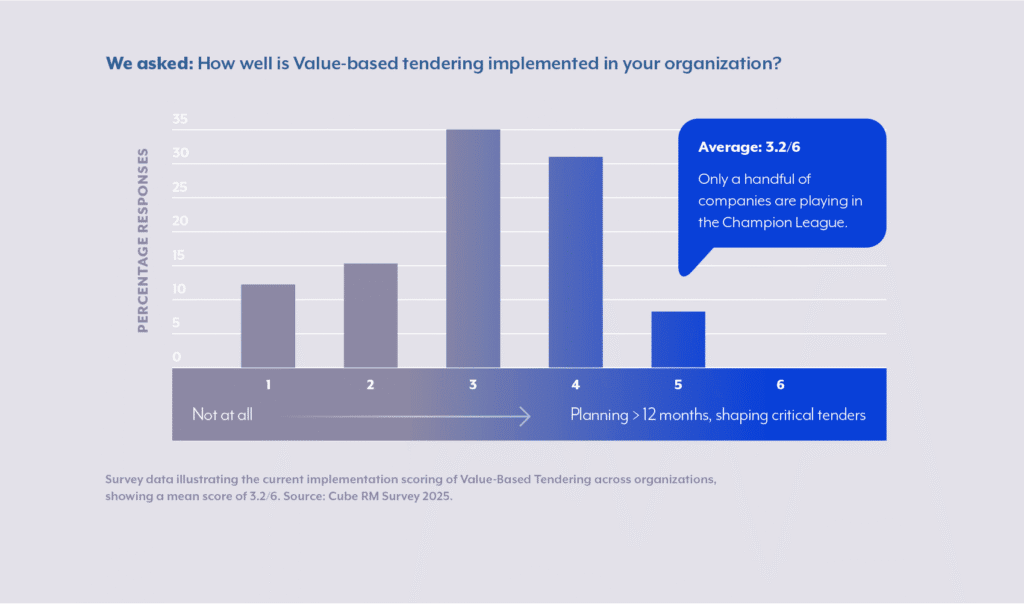
The foundation of effective value-based tendering lies in early identification and anticipation through a Tender Calendar for relevant tender opportunities. Currently, this represents a significant gap in most pharmaceutical and medical technology companies’ tender management processes. Cube RM’s recent survey of leading pharma companies revealed that around 25% of companies operate in hyper-reactive mode, typically learning about tender opportunities only 3-4 weeks before submission deadlines. Value-based tendering requires, amongst other things, pro-active planning, strategy definition, internal coordination across multiple functions, relationship building with external stakeholders and many more things, which all require adequate time. A reactive approach severely limits the ability to implement meaningful tender shaping strategies or develop compelling value propositions.
Successful value-based tendering demands a shift toward proactive tender anticipation, ideally 12-24 months before publication. This extended timeline allows organizations to engage with key stakeholders, gather necessary real-world evidence, and influence tender specifications before they become fixed requirements. Advanced technology solutions now enable this proactive approach through sophisticated forecasting algorithms that analyze historical tender patterns, contract expiration dates, and procurement cycles to predict when specific tender opportunities will be published and with what probability of occurrence. By leveraging these predictive capabilities, tender teams can transform from reactive participants to strategic shapers of their market opportunities.
LEVERAGING TECHNOLOGY FOR TENDER FORECASTING
Many market access and tender teams invest considerable resources manually scanning multiple platforms or relying on informal industry networks to predict when opportunities might emerge. This creates strategic blind spots that compromise competitive positioning. Without adequate lead time, organizations struggle to align internal teams, develop differentiated value propositions, or engage in pre-tender market shaping activities with external stakeholders.
Advanced forecasting solutions are emerging to address this critical gap. Tender intelligence platforms can now analyze historical procurement patterns, contract cycles, and publication behaviors across different contracting authorities to predict not just when tenders will be published, but with what level of confidence.
The strategic value of such forecasting capabilities extends beyond simple calendar planning. Leading-edge platforms like Cube RM’s Tender Central now incorporate sophisticated forecasting modules that provide predictions with confidence scores, enabling organizations to shift from reactive bid preparation to proactive market development. This transforms tender management from a defensive operation into a strategic competitive advantage, allowing life sciences companies to shape their market opportunities rather than simply respond to them.
Once organizations have established visibility into future tender opportunities through forecasting, the next critical step involves strategic prioritization. Not every anticipated tender justifies the intensive resources required for effective value-based tendering initiatives. Value-based tendering activities demand significant investment in stakeholder engagement, and prolonged market development efforts, often extending to 12-18 months of dedicated work across multiple departments.
The challenge lies in systematically identifying which opportunities justify this level of investment versus those better suited for standard competitive bidding approaches.
Effective prioritization requires a multidimensional evaluation framework that considers several key factors. Market dynamics play a crucial role: opportunities with limited competition or where organizations hold distinct clinical advantages present stronger candidates for value-based approaches. Product portfolio alignment is equally important, as tenders involving differentiated therapies typically offer greater optimization potential than generics. For Generics and Biosimilars, this requires focusing on differentiators beyond the molecule, such as supply reliability, packaging, services, ESG-criteria, storage conditions, return policies etc.
Geographic factors also influence prioritization, as some regions demonstrate greater receptivity to value-based procurement approaches than others.
TECHNOLOGY-ENABLED QUALIFICATION SYSTEMS
Advanced tender management platforms now incorporate automated qualification capabilities that streamline this complex prioritization process. Solutions like Cube RM’s Qualification Index within Tender Central enable organizations to establish customizable scoring frameworks based on their specific strategic criteria, products, budgets, award criteria etc.
These qualification systems automatically evaluate incoming opportunities against predetermined criteria, bases on a 5-star logic system that enables decisions while reducing the manual effort required to assess hundreds of potential opportunities. The next evolution of this process lies at the agentic level, where task-specific intelligent agents can be trained to identify and recommend the most strategic opportunities, a transition we explore further in our section on digital enablers. In this context, agents are rule- and data-constrained systems designed to augment human decision-making rather than replace it.
This systematic approach is needed to level-up tender prioritization from an ad-hoc discussion into a data-driven strategic process, enabling organizations to focus their most intensive optimization efforts where they will generate maximum business impact.
Together, these two dimensions establish the “where to play” and “how to play” framework of value-based tendering. Internal assessment ensures resources are focused on the most promising portfolio segments, while external archetypes reveal which tender environments are receptive to value-based propositions and where traditional price competition will still dominate.

VBT MARKET ARCHETYPES: DATA-DRIVEN TENDER MARKET LANDSCAPING
A data-driven market archetype framework represents another enabler of effective value-based tendering. At its core, a market archetype is a structured characterization of a specific tendering environment, that captures who the relevant tender authorities are, how they behave, how often tenders occur, what criteria are evaluated, and how competitive dynamics evolve over time. By building and maintaining these archetypes, companies can gain deep visibility into how tender markets function and change, essential intelligence for anticipating future dynamics and aligning tender strategies.
This approach combines quantitative and qualitative insights to create a multidimensional understanding of each market. Dimensions typically include: (1) tender market importance and size, helping teams measure strategic relevance and revenue potential (2) tender activity per market, which tracks frequency and intensity of procurement (3) procurement type (direct or indirect), indicating whether tenders are issued by hospitals, group purchasing organizations, or central authorities (4) award criteria and trends, distinguishing between MEAT-based markets that reward various qualitative criteria (e.g. supply, packaging, safety, environmental criteria etc) versus price-driven markets focused on cost minimization (5) tender duration, long-term versus short-term contracts and (6) winner structure, determining whether tenders tend to favor a single supplier or allow for multiple winners. (see image below)
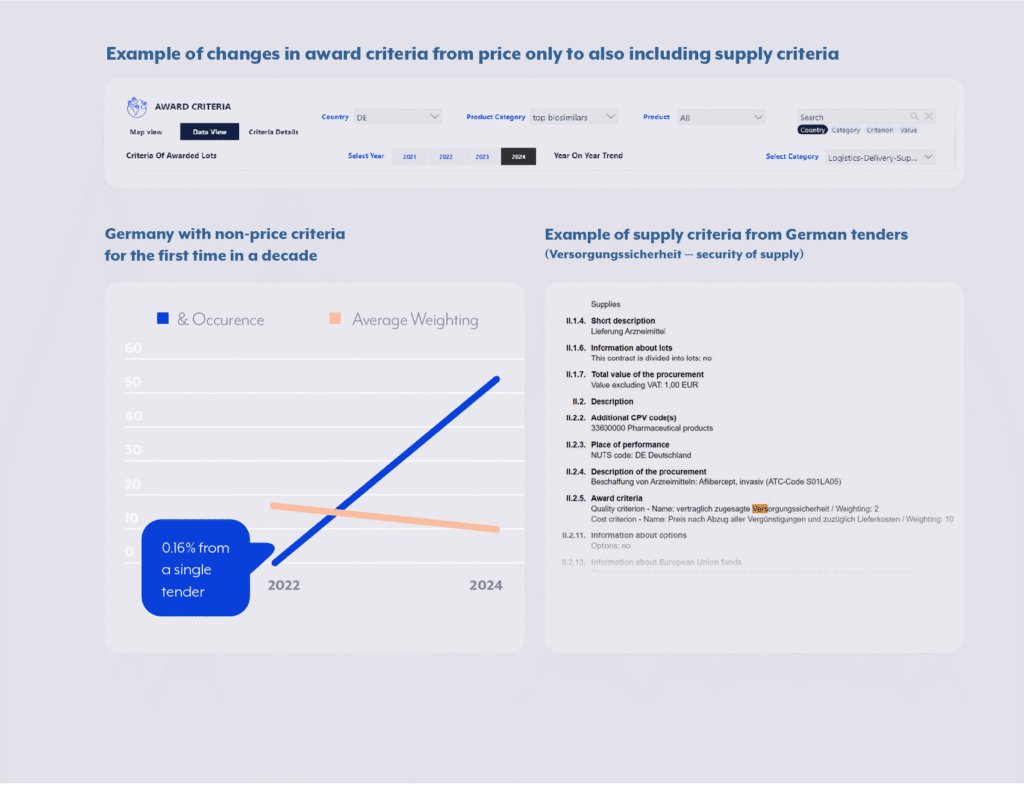
By quantifying these factors and continuously updating them, companies can classify countries, therapeutic areas, or contracting authorities into recognizable archetypes such as price-competitive, volume-driven markets versus innovation-oriented, outcome-based systems. This enables tender and market access teams to predefine playbooks for each archetype: where to emphasize clinical evidence, when to pursue collaborative partnerships, or how aggressively to compete on cost. More importantly, this structured view provides early warning signals when a market begins shifting from one archetype to another, for instance, when a previously price-dominated system starts adopting MEAT criteria or sustainability metrics.
e.g. Germany for many years, was a typical ‘price-only’ country, driven by big national insurance tenders. However, due to recent supply challenges and shortages, we can observe an inclusion of supply criteria in tenders in 2024–2025.
Over time, this continuous market intelligence becomes a strategic asset, a map of global tender behavior that guides resource allocation and the tailoring of value narratives.

Phase 2
Assessment & Selection
Once the targeted tenders have been identified in Phase 1, the focus shifts to the critical work of Concept Selection. This is where the strategy moves from theoretical to operational. It is not enough to simply identify a tender as a target; manufacturers must define how they intend to win it without eroding value elsewhere.
From our perspective, the traditional view of tendering, often limited to unit-price negotiations, is insufficient for Maximizing Tendering Outcomes through Successful Value-based-Tendering modern healthcare markets. Instead, quality criteria must be evaluated through a Product-Specific Lifecycle Lens. This holistic approach weighs overall health outcomes against total costs, ensuring that short-term price concessions do not undermine long-term asset value.
To rigorously qualify potential Value-Based Tendering (VBT) concepts, we recommend a dual-dimensional assessment framework: Attractiveness and Feasibility.
A concept must be desirable before it is actionable. We assess attractiveness by looking outward at the customer’s needs and inward at the manufacturer’s strategic imperatives.
ATTRACTIVENESS: EVALUATING VALUE FOR THE CUSTOMER AND THE MANUFACTURER
A concept must be desirable before it is actionable. We assess attractiveness by looking outward at the customer’s needs and inward at the manufacturer’s strategic imperatives.
A. External Attractiveness: The Customer View
The primary goal is to ensure the VBT concept resonates with the Tender Authority’s reality.
- Addressing Clinical & Budgetary Pain Points: A strong concept goes beyond the molecule. Does it solve a specific problem for the payer? For example, if a Tender Authority is struggling with budget unpredictability, a “capped cost” model may be attractive. If their concern is clinical uncertainty, a “pay-for-performance” model based on real-world evidence addresses that risk. The concept must directly answer the question: Does this alleviate a clinical bottleneck or a budgetary leak?
- Enhancing Therapy Value & Usage: The concept should augment the therapy itself. Highpoint emphasizes the role of the “total solution.” This includes leveraging medical devices or digital companions to increase patient compliance and adherence. By bundling the drug with adherence tools (e.g., smart injectors or app-based monitoring), manufacturers offer a guarantee of better outcomes, not just the delivery of doses.
Experience shows that many projects fail when the tender authority’s perspective, priorities and operational realities are not sufficiently considered. It is critical to first gain a deeper understanding of the buyer’s situation before building a value-based narrative and strategy.
INTERNAL ATTRACTIVENESS: THE BUSINESS VIEW
Equally important is whether the concept serves the broader strategic goals of the manufacturer.
- Strategic Compatibility: No tender exists in a vacuum. The selected concept must be compatible with the pricing and access strategies for future indications, pipeline products, and associated medical devices. We must ensure that a creative tender structure today does not set a precedent that hampers the launch of a follow-on indication tomorrow.
- Global Price Integrity & Competitive Advantage: Rather than engaging in a race to the bottom, our VBT concepts leverage our superior product profile and company services to justify value beyond the unit price. By bundling our market-leading support services, proprietary delivery technologies, and proven outcome data, we create a “value shield” that protects our international price floor. This allows us to offer competitive tender bids based on total value delivered rather than transparent discounts, preserving our global pricing power while outmaneuvering low-cost competitors who lack our comprehensive ecosystem.
FEASIBILITY: THE OPERATIONAL REALITY CHECK
The second dimension acts as a funnel: many concepts are attractive, but few are truly feasible. This assessment prevents the selection of concepts that look good on paper but fail in execution.
A. Stakeholder Alignment
- Corresponding Interests: A VBT concept often requires collaboration between the Payer (who pays) and the Provider (who prescribes/administers). A concept might appeal to a Tender Authority because it saves money, but if it increases the workload for hospital staff without compensation, it will fail. Feasibility requires mapping the concept against the interests of all stakeholders—providers, authorities, and patients—to ensure incentives are aligned.
B. Operational Complexity & Administrative Burden
- Manageable Complexity: This is the most common failure point for Value-Based Tenders. If a concept requires tracking complex patient data points that the hospital’s IT system cannot easily generate, the administrative burden will strangle the deal.
- Cost-to-Serve: We must assess the cost to set up, run, and assess the tender. Does the concept require hiring new field staff? Does it require purchasing third-party data? If the cost of administering the VBT erodes the margin gained by winning the tender, the concept is not viable. True feasibility means the concept operates within a manageable framework of complexity and cost.
SUMMARY: THE SELECTION MATRIX
By plotting potential concepts against these two dimensions—Attractiveness (High/Low) and Feasibility (High/Low) enables teams to objectively prioritize. We aim for the “Sweet Spot”: concepts that solve genuine customer problems, protect internal pricing, align with provider workflows, and can be executed without excessive administrative overhead.

Phase 3
Operationalization & Execution
After identifying the right markets, tenders, and value concepts in Phases 1 and 2, the third phase focuses on bringing this strategy to life and translating strategic intent into structured execution. This phase operationalizes the shaping plan ensuring that every insight, narrative, and stakeholder engagement is transformed into concrete and measurable action.
DEVELOP THE DIFFERENTIATION STRATEGY (BID-VALUE-TABLE)
At the heart of this phase lies the Tender-Value-Matrix, a structured methodology that connects customer objectives to the company’s differentiated value propositions. The process begins with a deep understanding of the customer’s objectives and challenges. This can be derived from RFPs, previous interactions, public documents, or KAM (Key Account Management) insights.
The Bid-Value Table guides teams through four key questions:
- What are the customer’s objectives and needs?
- How does our solution address these objectives?
- What tangible value and benefit does the customer gain?
- What credible evidence can we provide to substantiate our claims?
By systematically answering these questions, the organization creates a cohesive value narrative, one that clearly articulates how its offer aligns with the tendering authority’s priorities. This narrative becomes the foundation for the differentiation strategy, linking internal strengths to external customer goals in a way that is both evidence-based and compelling.
STAKEHOLDER MAPPING AND ENGAGEMENT
Understanding the customer means understanding its people. Successful tender optimization requires a detailed stakeholder mapping exercise, identifying who the key decision-makers and influencers are within the contracting authority. Stakeholder objectives can often be understood through public documents or professional networks, but the most powerful insights come from structured engagement, public information, prior interactions, or insights shared by Key Account Management (KAM) teams.
The stakeholder map should classify contacts based on their role (decider, influencer, technical evaluator, or procurement lead) and document their key interests and concerns. This enables personalized communication strategies and ensures that the value narrative resonates with each stakeholder group.
EXECUTION OF THE VBT PLAN
Once the differentiation strategy and stakeholder map are established, disciplined execution becomes critical. Using task and workflow management, teams can coordinate Value-Based activities through defined milestones, checklists, alerts, and progress tracking. This ensures that key activities, such as Bid-Value-Table preparation, internal reviews, and pre-tender alignment are completed on time and with accountability.
Because value-based tendering is inherently cross-functional, efficient collaboration is essential. Market Access, Public Affairs, Sales/KAM, Marketing, Medical, Legal, and Regulatory teams all contribute to building and validating the value proposition. A shared digital workspace can bring these stakeholders together, enabling real-time coordination, version control, and document management. Working from a single source of truth minimizes duplication, ensures compliance, and keeps strategic and operational objectives aligned.
DIGITAL ENABLERS: FROM TENDER MANAGEMENT TO INTELLIGENT ORCHESTRATION
The full potential of the execution phase is realized when these processes are supported by advanced digital tools. Modern tender management platforms consolidate data, people, and processes into a single environment, transforming tendering into a transparent, data-driven discipline where every step is traceable and strategically aligned.
As Agentic AI capabilities mature, this orchestration becomes increasingly structured and semi-autonomous, moving away from manual processes toward guided, system-supported execution. AI agents can assist across all key stages of the tender lifecycle by continuously scanning for upcoming opportunities, planning key activities of the tendering team well in advance and even drafting submission drafts. In this role, agents act as intelligent collaborators that augment human expertise, learning from company data, user feedback, and past tender results, while still following predefined rules. For tender and market access professionals, this means less time spent on administrative coordination and more on strategic decision-making. The result is an AI-augmented tender organization, where strategic shaping initiatives become both scalable and measurable.
Conclusion and outlook
Value-based tendering is a fundamental shift in how pharma and med-tech companies can win tenders in this era of increasing complexity and cost pressure. By anticipating opportunities, evaluating the full portfolio and services, and focusing on differentiated value, companies can move the competitive focus away from price-only decisions.
The value of VBT lies in its ability to align tendering with long-term strategic goals. Instead of spreading resources across every opportunity, companies can concentrate on tenders where they bring unique value, increasing win rates while safeguarding profitability. Moreover, value-based tendering fosters stronger, trust-based relationships with procurement authorities, who increasingly expect suppliers to deliver on more than just cost savings.
That said, value-based tendering is not without its limitations. It requires significant internal alignment, high-quality data, and disciplined prioritization. There is also the risk of over-engineering strategies where procurement bodies remain primarily price-driven. Companies must therefore balance ambition with practicality, ensuring that efforts to differentiate on value do not drain resources from tenders where price is still the decisive factor.
Looking ahead, the role of Agentic AI in value-based tendering could be transformative. The software industry is increasingly moving toward agentic computing, systems in which task-specific AI agents operate within defined rules, data boundaries, and workflows to support complex processes. Applied to tendering, this could mean agents that automatically anticipate VBT-worthy upcoming opportunities, continuously monitor contracting authorities, competitors, and historical outcomes, and then autonomously suggest shaping strategies for specific tenders.
However, it is critical to understand what agentic AI really means in practice. This is not about deploying a one-off AI tool or a single “super-agent” to solve all challenges. That would be like hiring a brilliant employee and burning them out with impossible expectations. Instead, multiple, domain-specific AI agents must be embedded into existing workflows and applications, learning iteratively from company data and user feedback Working alongside people rather than replacing them, they form a new kind of digital workforce that amplifies the capabilities of tender and market access teams.
For example, a Value-Based Tendering Agent can continuously screen upcoming opportunities that qualify for shaping, those that are significant in terms of revenue and profit-expectations and scheduled far enough in the future to allow time for preparation and all the needed activities. The agent could also learn based on other criteria such as whether a company won or lost the previous version of the tender, the number of expected competitors or the award criteria with their respective weightings.
As a next step the Value-Based Tendering Agent can design a recommendation for a strategic shaping plan: competitor analysis, portfolio positioning, risk assessment, and value narrative recommendations. Users can feed the agent with relevant inputs and documents such as Value dossiers, SMPC (Summaries of product characteristics) or latest clinical study results in order to refine the narrative and strengthen the value proposition.
Because value-based tendering is inherently a cross- functional effort involving multiple departments, the agent can also act as a coordinator: alerting, reminding, and guiding colleagues on their respective tasks, ensuring smooth collaboration and timely deliverables.
However, just as in other industries, companies must be cautious of the hype cycle. Agentic AI is powerful, but widespread adoption in tendering may take some time, and profitability gains will depend on a step-by-step and strategic implementation.
According to the MIT report ‘State of AI in Business 2025,’ the failure rate for generative and agentic AI projects remains very high. However, successful initiatives with positive ROI impact consistently share common traits: meaningful integration into existing processes workflows and software, high domain focus / expertise, and iterative human-AI collaboration.
Another important element is the quality of the underlying body of data used to train the agent. Without high quality data even the most sophisticated model will fail do deliver.
In this context, organizations like Cube RM leverage high domain fluency and focus in healthcare alongside software and process understanding and, in many regions, over 5 years of historical public tender data to meet these standards.
Agentic AI systems are only as effective and helpful as the quality and transparency of their reasoning and recommendations. In the healthcare and life sciences context, the tolerance for inaccuracies or hallucinations is extremely low. Therefore, companies developing or deploying AI agents must deeply understand the healthcare setting, its responsibilities and complexities, and train or structure their agents accordingly. In addition to that, all agent outputs should be traceable, logged, and auditable, allowing organizations to review inputs, reasoning steps, and outcomes retrospectively.
In particular, any statement with a medical implication must always be evidence-based, and citable. Every recommendation should follow a transparent logic chain of challenge/question > evidence > conclusion to ensure the reasoning process can be trusted. Above all, strong human oversight will remain indispensable.
At Cube RM and Marbls, we believe the future lies in this collaborative model: human expertise guided by intelligent agents that adapt, learn and scale. With this approach, value-based tendering becomes more than a strategy, it is a practical path, that ensures pharma and med-tech companies can win more tenders, at better margins, with stronger long-term impact.

About the Authors

Cube RM is the leading global Tender Software, AI & Data company, empowering Life Sciences organizations to find more business opportunities and build market intelligence. Our platforms use the latest AI, NLP, and Agentic AI technology, through Tender Agents specifically designed for Life Sciences tendering, to capture key tender market data globally and transform it into actionable market intelligence. Combined with seamless software automation and industry expertise, Cube RM helps your business increase tender process and tender teams efficiency.
Marbls partners with pharmaceutical and medical device and diagnostic manufacturers to provide practical, innovative services that address complex pricing, contracting and commercial excellence needs — driving the path of critical drugs, devices, and diagnostics to market. We invest in the long-term success of our clients by investing in our employees. As a team with world-class experience and a client-first philosophy, we create long term relationships based on success, service and trust. Our approach is results driven, hands-on and flexible to address the unique internal and external dynamics impacting our clients.
References
- Cube RM Survey + Tender Status Report 2025
- European Federation of Pharmaceutical Industries and Associations (EFPIA)
- MIT report ‘State of AI in Business 2025